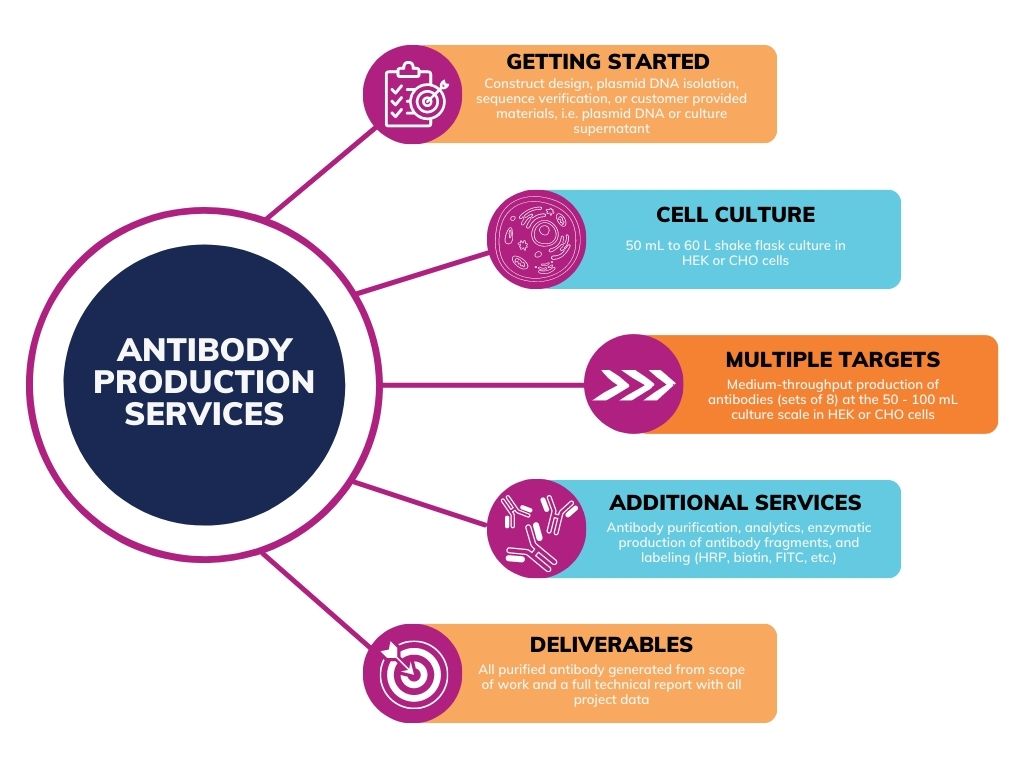
Antibody Production
Recombinant expression and purification of full-length IgG and antibody fusion proteins. Our comprehensive production capabilities encompass construct design, transient mammalian cell expression using in-house or customer-provided expression vectors, and affinity purification.